The fact that hardness of water varies throughout the US. This makes domestic water users concerned about how it would affect their health and life quality. What are hard water buildup problems and solutions?
In today's article, we will clarify these inquiries.
FYI: What Is Hard Water?
Hard water is commonly found in those water systems using groundwater. When moving through aquifers, it dissolves a variety of metals, especially compounds of magnesium and calcium.
And, when the content of calcium carbonate exceeds 120 mg/L (milligrams per liter), it’s classified as hard water.
Hard water, in general, causes many side effects and inconvenience to not only your daily life but also your health.
Harms of Hard Water Buildup
On Human Health
Showering with hard water every day causes dry skin, dry hair, or rashes.
The dissolved minerals in hard water change the skin’s pH balance, which is extremely bad for eczema sufferers.
Drinking hard water for a long time, minerals contained in the water easily cause kidney stones, urinary stones, or clog the inner walls of arteries and veins, which is dangerous to human health.

According to some studies, both heart diseases and high blood pressure can be caused by drinking hard water.
On daily Life
Calcium in hard water reacts with cleaning solutions, which creates soap scum. That explains why there is significantly less foam than usual when dissolving soap or cleaning detergents in hard water.
Besides, after washing your hands or glass items, you'll often see a thin film left on the surface.

To cleanse off that film, more cleaning fluid is needed.
Washing clothes in hard water for a long time causes the fabric to deteriorate and fade. Not to say that, after washing, stains or undissolved soap powder are found on them.

Hard water causes caulking on metal items, such as faucets and showerheads – either they will get rusted or appear white spots.

Moreover, it might clog water pipes in your house as well as create yellow stains on many household items, which costs you more money for pipe repairs or replacement.
Raw foods that are cooked in hard water need more time to be done. Moreover, their tastes and flavors are hugely decreased.

When making coffee or tea with hard water, these drinks will be darker, less fragrant, and taste pale.
In Industry
In textile industry
Hard water greatly affects the dyeing, washing, and finishing process.

Particularly in the washing section, it causes the fibers to stiffen and scratch. Hard water with high content of iron creates a reddish-yellow color on washed fabrics.
In addition, as soap can’t be dissolved in hard water, textile products aren’t washed thoroughly, resulting in residue and dirt still adheres to the fabric. This makes product quality decrease noticeably.
In the beverage industry
In the beverage industry, the quality of water sources is important. They need to be safe, and free of contaminants and microorganisms.
If unfortunately, hard water is used, it will change the product’s color and taste. Moreover, businesses will have to pay a high amount of money to reduce and soften the water hardness.
Impacts on industrial-grade boiling and cooling equipment
Water, in this equipment, is used as the heat transfer medium.
If the water source has high hardness, it might cause corrosion, calking buildup, and clogging. If the boiling and cooling equipment is heavily clogged, it is potentially explosive due to high pressure.
Calking buildup in the boiler reduces the tool’s operating efficiency because they have to consume more electricity.
Not to say that hard water shortens their lifespan.
Solutions
Hard water is divided into two types:
- Temporary hard water - caused by Ca(HCO3)2 and Mg(HCO3)2 salts
- Permanent hard water - caused by salts of MgCl2, CaCl2, MgSO4, CaSO4
The solid principle in softening (all types of) hard water is to reduce the concentration of Ca2+ and Mg2+ ions in it.
Precipitation Method
With temporary hard water
Let it boil for several minutes to make sure all the minerals are eliminated completely. For better results, add a reasonable amount of Ca(OH)2 or other alkaline solutions, such as CO32 and PO43.

With permanent hard water
Add CO32 and PO43.
White Vinegar

Chemically, calcium is alkaline, meaning that its pH level is greater than 7. White vinegar, on the other hand, is very acidic and its pH level is around 2.5. Therefore, it can help neutralize the calcium content in hard water.
However, you wouldn't want to pour a whole bunch of white vinegar into your drinking water, would you? That's why this method is only ideal for cleanings, such as clothes-washing or cleaning stains on metal appliances.

Ion Exchange Method
This is the most popular and optimal method because of its low cost.
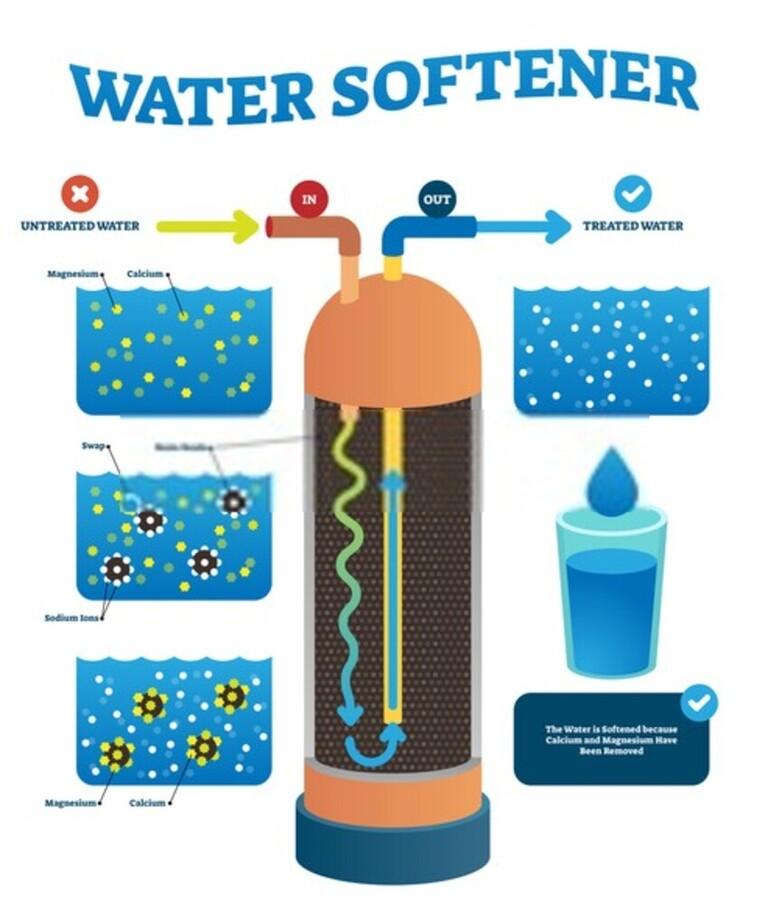
The operating principle of this method is to use ion exchange resin to replace harmful free ions in the water to form harmless substances.
Reverse Osmosis (RO) Systems
This method can eliminate almost all minerals and contaminants in hard water by forcing them through a filter built with a semi-permeable membrane.

The calcium and magnesium residue retained by the filter will be pushed out along the wastewater stream.
Conclusion
Above is all the basic information we’d like to share with you. Hopefully, it gives you an overview of hard water buildup problems and solutions.
Thank you for reading and if you have any questions about this topic, do not hesitate to leave a comment right below, we’ll try to respond as soon as possible.













